ADULT BRAIN TUMORS
The most aggressive and often fatal brain tumor is glioblastoma multiforme (GBM).
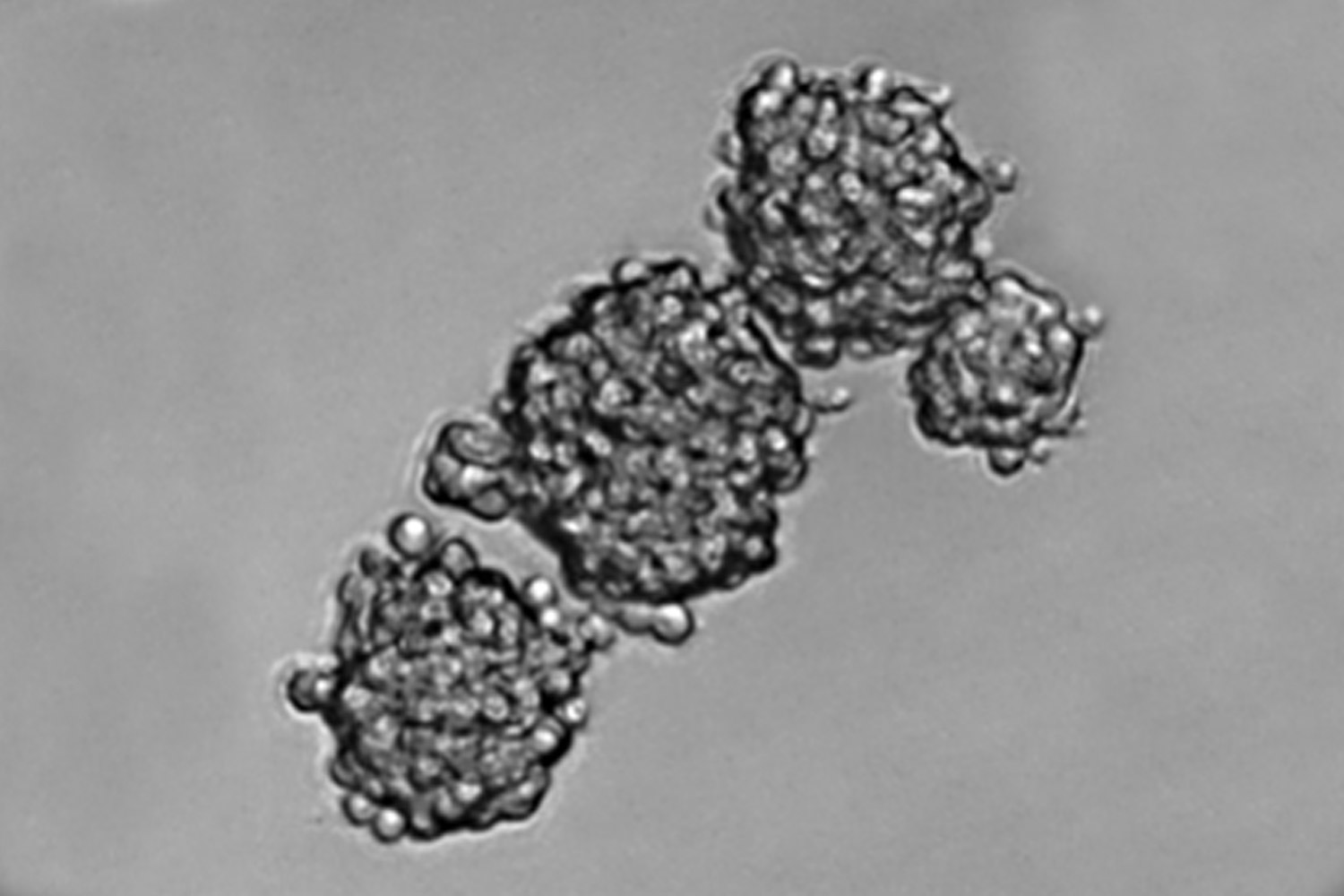
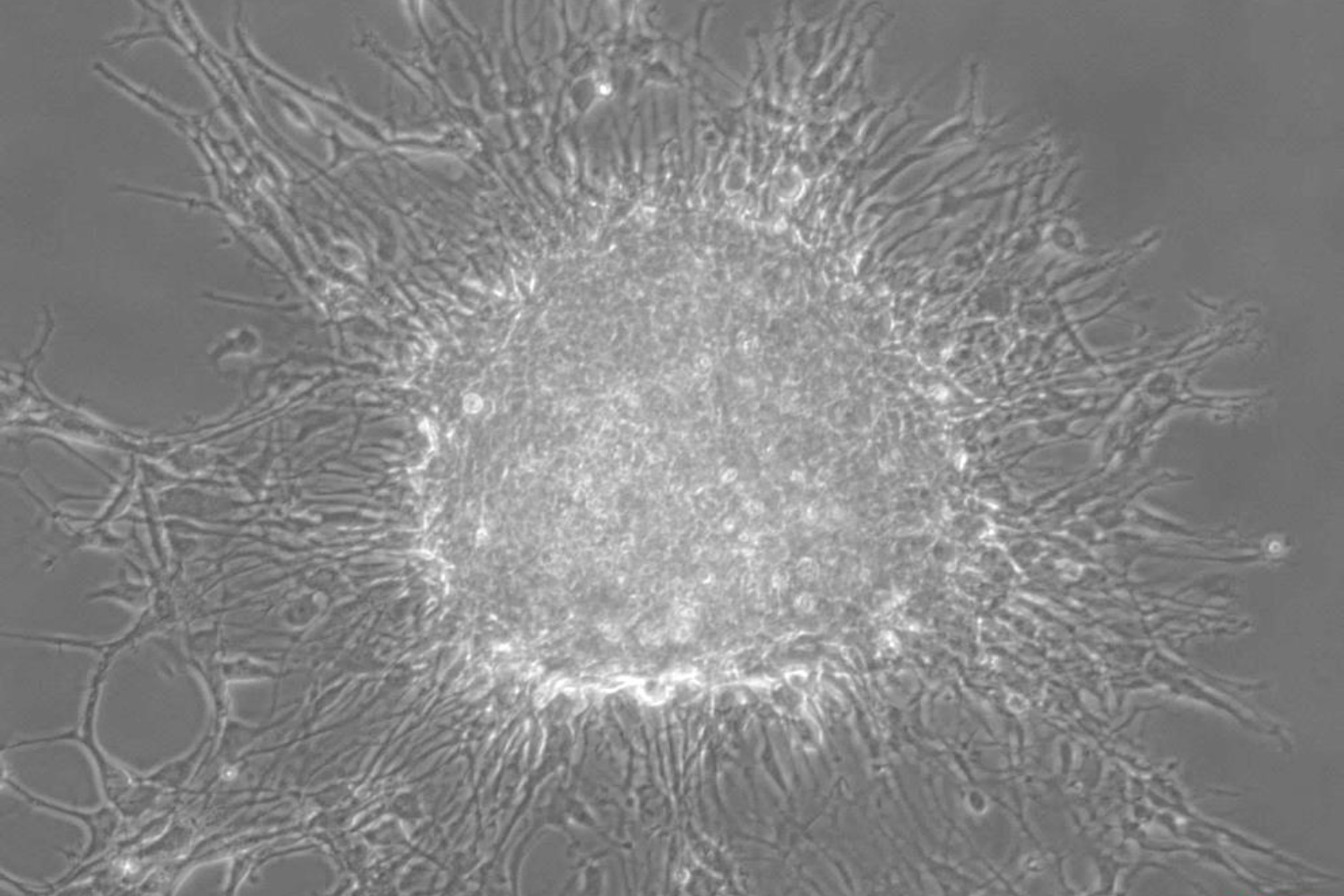
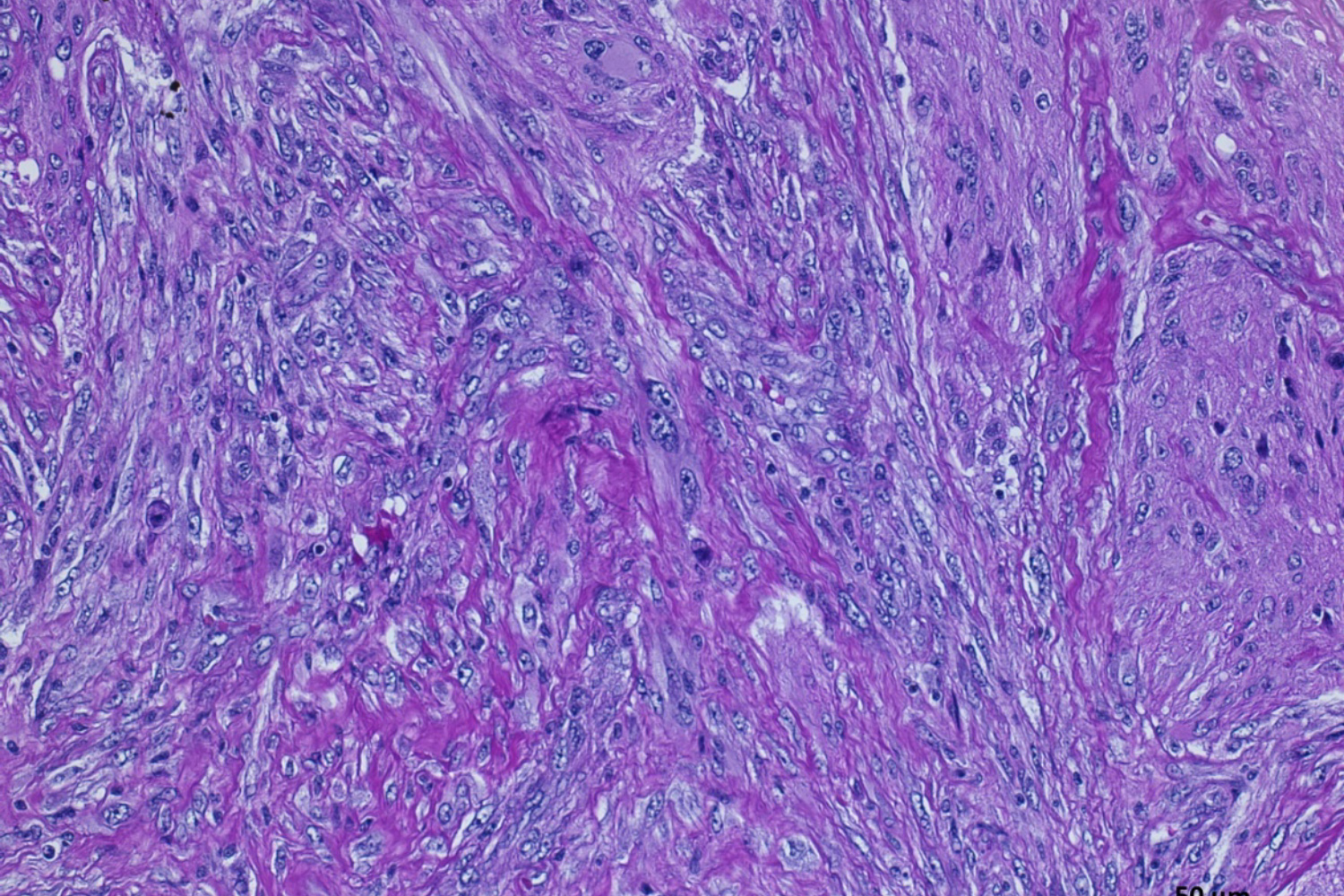
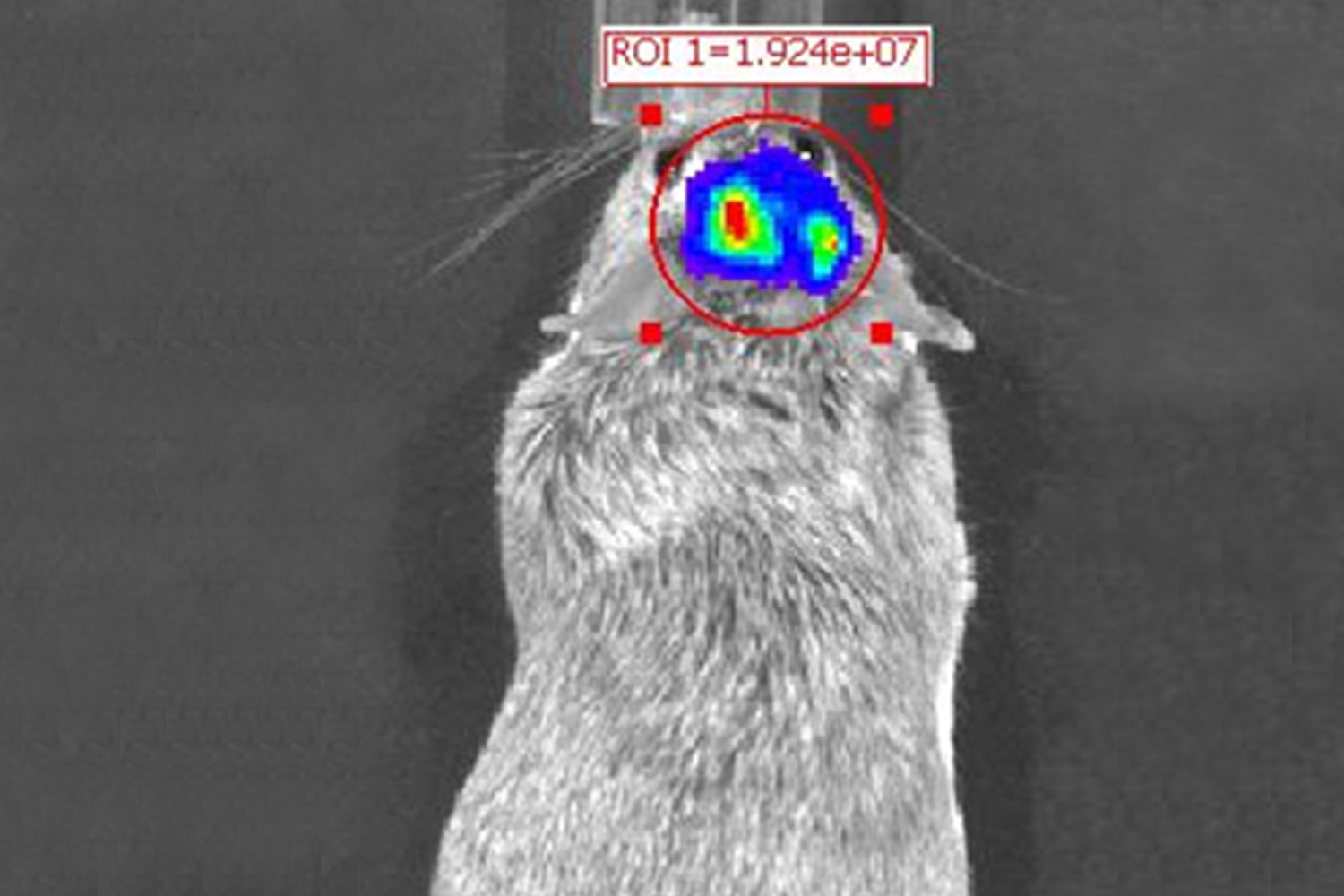
Our translational research program focuses on malignant primary adult brain tumors, including GBM and tumors of the brain meninges (meningioma). To better understand tumor biology and test novel treatment strategies, we have generated several novel cell models from patient-derived primary brain tumor tissues for our molecular and proteomic studies. We continuously collect blood samples from GBM patients and use these clinical samples in “omics” studies for the discovery of peripheral and metabolic biomarkers. Patient-derived GBM initiating cells are used for GBM targeted drug tests and injected into mouse brain to assess in-vivo treatment responses.
Dr. Klonisch is the Brain Tumor Lead in Manitoba of the GBM biobanking and research program for the Canadian Prairies co-funded by the Terry Fox Research Institute (TFRI) and CancerCare Manitoba
MAJOR TOPICS WE ARE CURRENTLY WORKING ON INCLUDE:
LITT is a minimally invasive hyperthermia-based cytoreductive method approved by the FDA for the treatment of inoperable brain tumors. We use a pre-clinical murine glioma model to evaluate the physiological mechanisms of LITT treatment on cerebral tissue in order to bridge knowledge gaps on the mechanisms of action of this emerging therapy. We use various imaging and staining methods, including the most sophisticated multiplex immunofluorescence, to understand the underlying repair mechanisms and systemic post-LITT effects.
Overcoming the blood brain barrier and achieving effective therapeutic drug levels at the brain tumor site are currently critical limitations for the treatment of primary brain tumors and brain metastases. We use in-silico designed brain permeable small allosteric inhibitors that are highly selective for ABL1/2 kinases. We test the efficacy of these novel drugs in vitro by using patient-derived brain tumor stem cells and in vivo by employing our brain xenograft mouse model. We also use novel compounds capable of reprogramming macrophages in the tumor microenvironment as a new therapeutic approach.
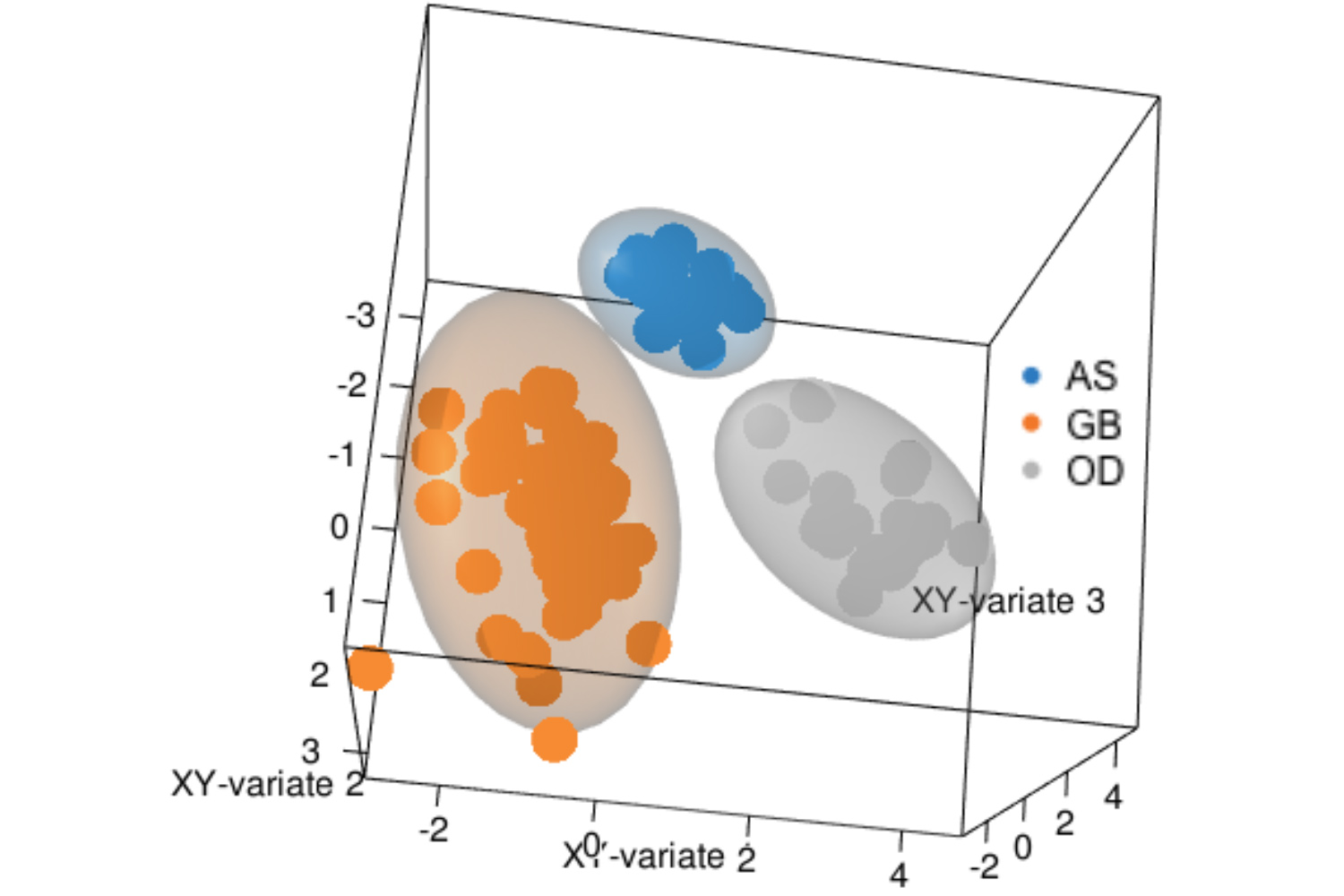
Proteins execute cellular functions and alterations in protein profiles directly affect cellular functions. We performed proteomic profiling on our patient-derived brain tumor cells and plasma samples to identify specific proteomic clusters in glioblastoma. Our bioinformatic analysis has identified actionable signaling pathways that are interrogating with selected drugs. Post-translational changes modulate protein functions and serve as novel biomarkers in our glioblastoma blood studies.
A substantial portion of a glioblastoma tumor mass consists of non-tumor resident cells and cells of the innate and adaptive immune system that have invaded the tumor. Our lab uses new compounds shown to reprogram innate immune cells to fundamentally disrupt the ability of the tumor microenvironment to support glioblastoma cell growth and survival. This exciting project is done in collaboration with the NIH and other brain tumor centers in Canada.