Publications by Dr. Thomas Klonisch
In his translational Oncology research projects, Dr. Thomas Klonisch mainly focuses on mechanistic studies to better understand therapeutic resistance in tumors, with a particular emphasis on Brain Tumors. His published work includes receptor signaling events affecting cell radio-/chemo-resistance and cell plasticity (EMT, stemness), drug nanocarriers, ER stress and unfolded protein response (UPR), cell death mechanisms (apoptosis, autophagy), and the effect of novel therapeutics on cancer (stem) cells and innate immune cells in the tumor micro-environment.
Publications by Dr. Sabine Hombach-Klonisch
The cancer research program of Dr. Hombach-Klonisch focuses on the cellular mechanisms that drive late stage cancer progression and therapeutic resistance in glioblastoma and in brain metastatic breast cancer. Her interest is to identify molecular mechanisms and cellular signaling events that promote cancer cell survival under cellular stress, including DNA stress and endoplasmic reticulum stress, which can be targeted to improve treatment success. She investigates the influence of brain resident cells of the perivascular niche, such as pericytes and brain endothelial cells, on these cancer cell stress responses.
A4: Mechanisms of therapeutic resistance in cancer (stem) cells with emphasis on thyroid cancer cells.
REVIEW
This review highlights mechanisms of therapy resistance in heterogeneous solid tumors that are linked to epithelial-mesenchymal transition, DNA repair capabilities, autophagy and endoplasmic reticulum stress.
A5: Role of cell death mechanisms and unfolded protein response in glioblastoma (GBM) chemoresistance
REVIEW
In this review, we summarize the impact of DNA alkylating drugs, like the GBM standard treatment drug Temozolomide, on resistance mechanisms initiated by GBM, with a particular emphasis on cell death mechanisms, endoplasmic reticulum stress and unfolded protein responses.
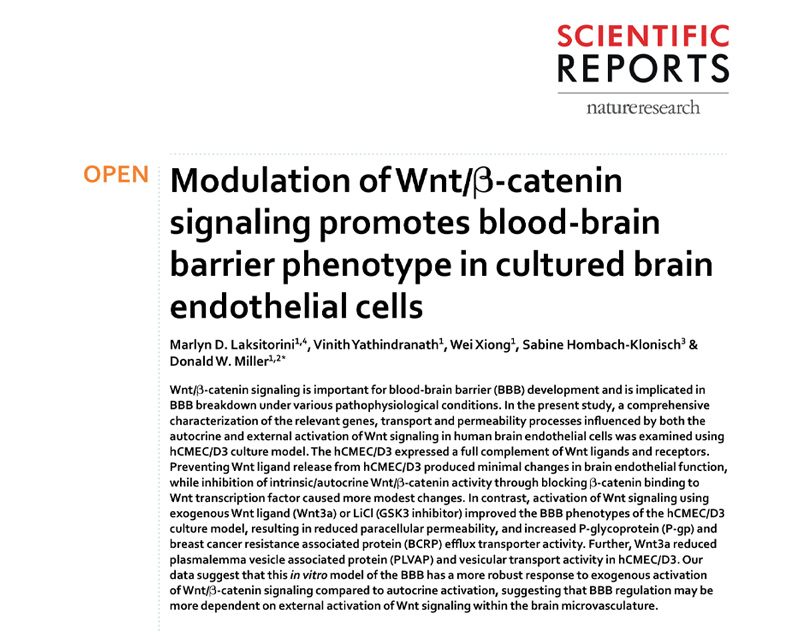
A6: Modulation of Wnt/β-catenin signaling promotes blood-brain barrier phenotype in cultured brain endothelial cells
KEY FINDINGS
This work was done in collaboration with Dr. Don Miller, Dept. of Pharmacology, University of Manitoba.
A7: A novel microfluidics device to monitor and quantify chemotaxis of tumor cells
KEY FINDINGS
This work is done with our collaborator Dr. Francis Lin, Professor, Dept. of Physics and Astronomy, University of Manitoba. The new radial microfluidics device can run eight chemotaxis experiments simultaneously with monitoring and quantification at the single cell level.
A8: Unlocking bone for proteomic analysis
KEY FINDINGS
This work was done in collaboration with Dr. Claudius Mueller and Dr. Virginia Espina, George Mason University, Fairfax, Virginia, USA.